Bentley, a Germany-based company and a global leader in minimally invasive medical devices for vascular treatments, had, in September 2022, acquired the rights of the GoBack catheter from Israel-based Upstream Peripheral Technologies.
After the acquisition, it rebranded GoBack as the BeBack in synchronization with its product portfolio which includes BeGraft coronary, BeGraft peripheral: 1st & 2nd generation, BeSmooth peripheral, BeGraft aortic, BeGraft PLUS, and BeYond.
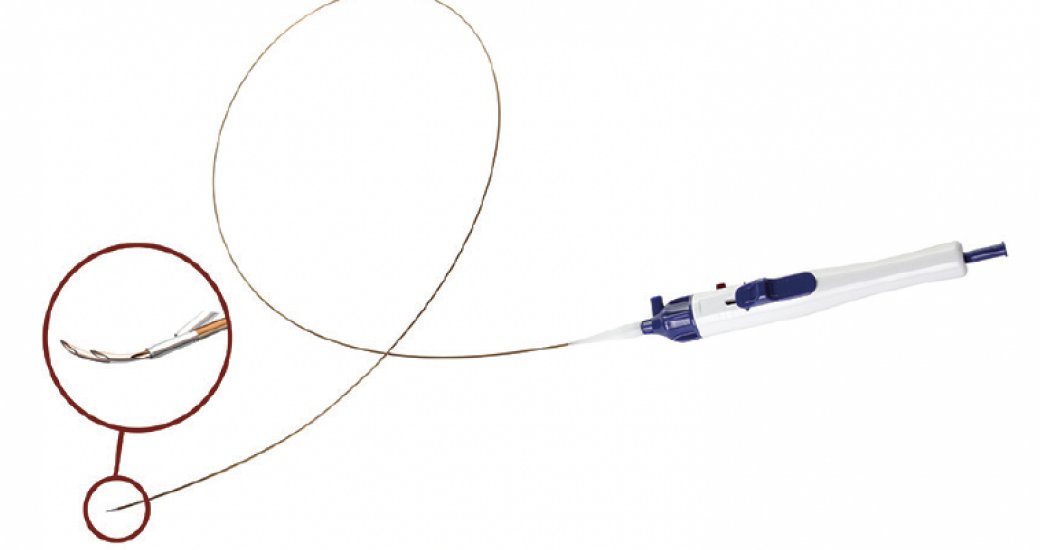
The BeBack crossing catheter is designed for the treatment of heavily calcified lesions. peBentley is inclined to make BeBack globally available in countries where the company is already active. The BeBack has received CE Mark approval and FDA clearance, making it easier to penetrate the European and the US market.
The BeBack crossing catheter is available in both an 80- and 120-cm catheter in both 2.9- and 4-F sizes. It has a unique steerable and adjustable nitinol (nickel-titan-alloy) needle, which can be easily manipulated by the physician. Also, a radiopaque marker indicates the direction the curved needle protrudes from the catheter tip.
The catheter allows for multiple treatment options including antegrade, retrograde, and crossover for the intraluminal or subintimal approach of stenotic or occluded lesions.
The device is currently being produced in Israel and shipped from there. Transfer of production to Bentley will be completed by 2025.










